If an amine compound has more than three carbon atoms, it will be available in the solid-state. Amines with a lesser number of carbon atoms are typically gases and have a fishy odour while amines with upto three carbon atoms are in liquid form. Amine compounds can exist in different forms depending on the number of carbon atoms present. However, if there are more carbon atoms present in an amine compound, then the solubility in water will decrease. Being hydrogen-based compounds, amines have elevated boiling points and a substantial degree of solubility in water. To gauge the extent of the uses of amines, let us first assess the physical properties of amines. The uses of anime majorly stem from the structure as well as the properties of amines. The structure and propensity to replace an electron make it appropriate for a wide range of uses of amines. If the substituent alkyls belong to the same group, then such amines are called simple and if it is constituent of more than one group, then they are called mixed amines. Secondary and tertiary amines can further be divided into cyclic amines when the compound forms a ring structure. Finally, tertiary amines are those whose hydrogen atoms have been replaced by an organic substitute. When two of the hydrogen atoms are substituted and one hydrogen atom bonded with a nitrogen atom, such amines are called secondary amines. When one of the three hydrogen atoms are replaced by an alkyl or an aromatic element, the amines are called primary amines. Depending on their bonding environments, amines can further be categorised in three ways. Fundamentally, the structure of amines comprise trivalent nitrogen atoms with an unshared pair of electrons. In general, the compounds of the amine groups are less reactive than the other organic compounds by virtue of their electron donating attribute. Some groups of amines also form an aromatic structure which in turn reduces their alkalinity. This salient structure of amines is to a large extent responsible for the multifaceted uses of amines. 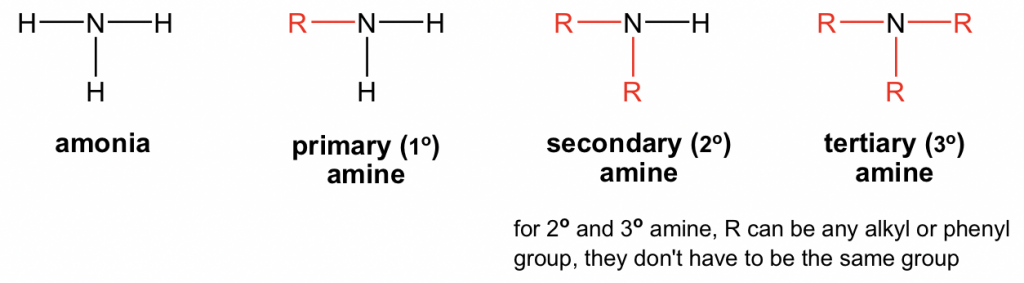 
As such, Amines have the following structure: Amines are compounds of the nitrogen group which are attached to a carbonyl within the structure. Amines compounds mainly form hydrogen bonds as a consequence of which they are highly soluble in water and have high boiling points. 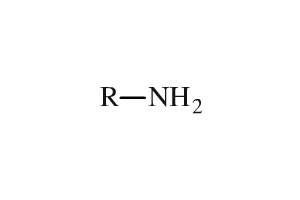
But, before we delve into the uses of amines, let’s understand what amines are and familiarise ourselves with their structural properties.Īn organic compound, belonging to the functional group comprising a basic nitrogen atom with a lone pair, are called amines. There are many uses of amines in our everyday lives. Be it the agrochemicals industries or the pharmaceuticals markets, amines are integral to the core as well as the by-products of these industries. As a derivative of ammonia, amines serve several practical purposes across different industries. Amines are derived by replacing one or more Hydrogen molecules from ammonia with one or the other alkyl. This includes examples of branches like health, energy, drugs, metals, textiles, paper, cosmetics, cleaners etc.Īmines are one of the most prolific members of the nitrogen-based organic compounds which also constitutes the indirect source of ammonia. Apart from the educational importance of this subject, Chemistry also actively participates in the everyday life of human beings. In this article, Students will learn a topic from the Chemistry stream, that is, Amines.Ĭhemistry is the branch of Science that deals with substances, their chemical properties and reactions. This includes physics, Chemistry and Biology streams of Science. For students, this subject deals with three main categories. This deals in every area of the universe. Science is a beautiful subject that studies the Universe with experimentation and trial and error. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
